Inspire posts forward medical research in adverse drug reactions, new study shows

Over 2 million injuries, hospitalizations and deaths in the US each year are associated with post-approval adverse drug reactions (ADRs).1 The usual method of gathering ADR data is through provider reporting. Research suggests that health care providers tend to under-report ADRs, sometimes because patients just don’t discuss all of their treatment experiences with them.2
Supplementing spontaneous provider-reliant reporting could be extremely beneficial to patient safety. In Can Patient-Generated Health Communication on the Internet Enhance Pharmacovigilance? (Part 1)3 and Research Confirms: Valuable Patient-Centered Data on Inspire (Part 2),4 I discussed the potential for life saving information on ADRs and other side effects from treatment concealed in the advice, support, and helping discussions among thousands of patients and caregivers on Inspire and how this information could be used to augment pharmacovigilance.
This idea is closer to reality now, with some demonstrable impacts for patients. Utilizing advanced data mining techniques and natural language processing, a research collaboration between Inspire and Stanford University School of Medicine uncovered a previously unidentified adverse reaction to chemotherapy. The findings, published in a Research Letter in the Journal of the American Medical Association (JAMA-Oncology) on March 1, also showed that ADRs may be reported on social media months earlier than through traditional methods.5
“Patients talk about their experiences in different places, not just in the ten minutes they get to spend with their doctor,” said Erik Jones, VP of User Experience and Data Science at Inspire and one of the co-authors*. “They’re in a different mindset when they’re on an anonymous Internet forum, often thinking of things that did not occur to them in a more formal setting.”
The team discovered that some patients receiving the chemotherapy drug erlotinib (Tarceva) reported hypohidrosis, which is the inability to sweat, a condition that can lead to heat exhaustion, heat stroke, or even death. This ADR had never been reported in the medical literature. Inspire members had been discussing it for over 11 years.
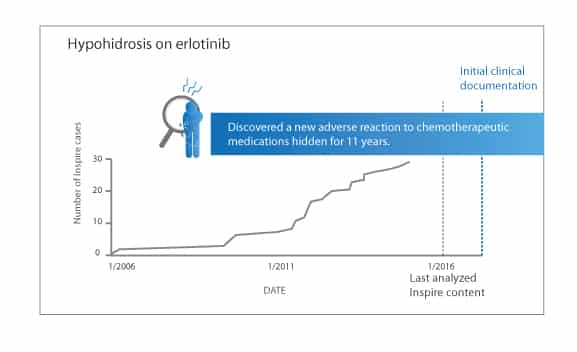
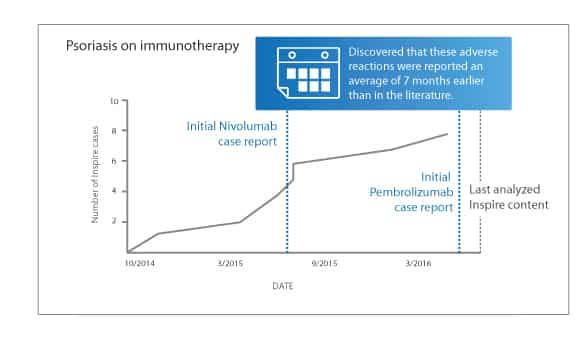
The team also found Inspire members were discussing ADRs much earlier amongst themselves – an average of 7 months before any of these side effects had been reported in medical literature.
Excavating social media posts
The researchers used an open source machine learning tool called DeepHealthMiner** that, together with natural language processing, helped them sort through millions of anonymized Inspire posts to look for associations between chemotherapeutic agents and ADRs.
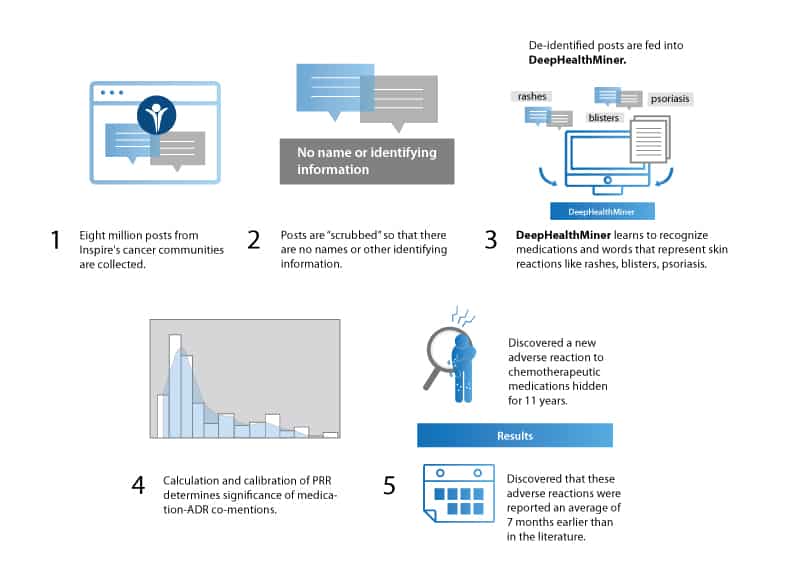
They focused on erlotinib (sold as Tarceva by Genetech), nivolumab (sold as Keytruda by Merck) and pembrolizumab (sold as Opdivo by Bristol-Myers Squibb) and adverse skin reactions to the medications like rashes, blisters, and psoriasis flares.
Calculating the proportional reporting ratio (PPR) helped the researchers determine how strongly a medication was associated with an adverse event. The higher the ratio, the stronger the association – enough, in some cases, to say a certain medication might be the cause of that ADR.
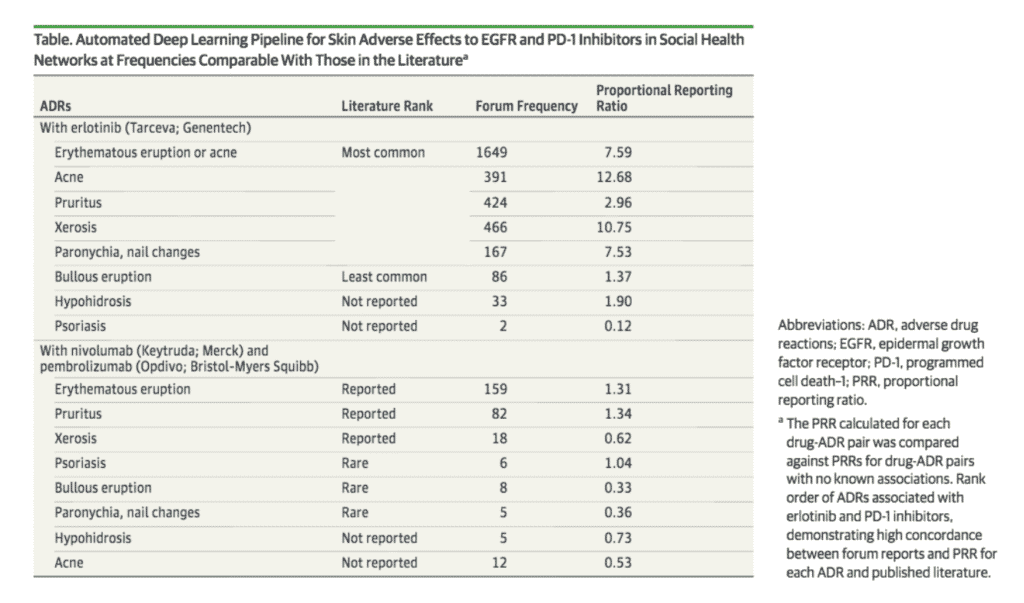
Detecting Chemotherapeutic Skin Adverse Reactions in Social Health Networks Using Deep Learning,” in JAMA Oncology. Published online March 1, 2018. doi:10.1001/jamaoncol.2017.5688]
Conclusions
If patients are reluctant or forget to report ADRs to their physicians, post-market vigilance is compromised. This information is essential to get to drug makers and regulatory bodies like the FDA early in order to warn other patients in advance of possibly deadly side effects.
Data mining social media discussions and natural language processing can significantly help patients, caregivers, and healthcare providers be aware of all possible ADRs earlier than they were before, and maybe reduce the number of injuries or hospitalizations and deaths that occur each year because of them. And, it’s another way to bring the patient voice to the forefront of research and care.
“I don’t think this is an exaggeration to call this Inspire’s most important contribution to advancing medical research to date,” Jones said.
* Researchers involved in the study:
Julia D. Ransohoff, AB, Stanford School of Medicine, Department of Dermatology,
Azadeh Nikfarjam, PhD Stanford School of Medicine, Division of Biomedical Informatics Research
Erik Jones, PhD, Inspire
Brian Loew, AB, Inspire
Bernice Y. Kwong, MD, Stanford School of Medicine, Department of Dermatology,
Kavita Y. Sarin, MD, PhD, Stanford School of Medicine, Department of Dermatology,
Nigam H. Shah, MBBS, PhD Stanford School of Medicine, Division of Biomedical Informatics Research
**Co-author Azadeh Nikfarjam, PhD developed DeepHealthMiner
Inspire offers a trusted community to patients and caregivers. Our goal with this blog, this website and our content is to provide the life science industry access to the true, authentic patient voice. In so doing, we support faithful operationalization of patient-centricity. Take a look at our case studies, eBooks and news outlet coverage.
References:
1 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3675775/
2 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4041129/
3 https://corp.inspire.com/patient-engagement/pharmacovigilance/
4 https://corp.inspire.com/patient-engagement/valuable-patient-centered-data/
5 https://jamanetwork.com/journals/jamaoncology/article-abstract/2673831